How to Define Reducing Agent and Its Role in Textile Dyeing
Table of Contents
In the sophisticated realm of textile chemistry, the ability to manipulate molecular bonds is what defines the quality of a finished garment. To define reducing agent in this context is to understand the fundamental catalyst that allows sulfur dyes and vat dyes to become soluble and penetrate fabric fibers effectively. Without these critical components, the vibrant colors and durable finishes we expect from modern apparel would be nearly impossible to achieve.
Globally, the textile auxiliaries market is evolving rapidly, with an increasing emphasis on precision and sustainability. Understanding how to properly define reducing agent applications helps manufacturers reduce chemical waste and energy consumption during the dyeing process. As global standards like ISO and ZDHC (Zero Discharge of Hazardous Chemicals) become more stringent, the role of efficient reduction chemistry has shifted from a simple industrial necessity to a core component of environmental stewardship.
For professionals in the dyeing and printing sector, mastering the chemistry of reduction is not just about technical compliance, but about commercial viability. By optimizing the use of reducing agents, factories can achieve higher color consistency, reduce the rate of fabric degradation, and lower overall production costs. This guide provides a comprehensive deep dive into the mechanisms, applications, and future trends of these essential chemical tools.

The Fundamental Chemistry of Reducing Agents
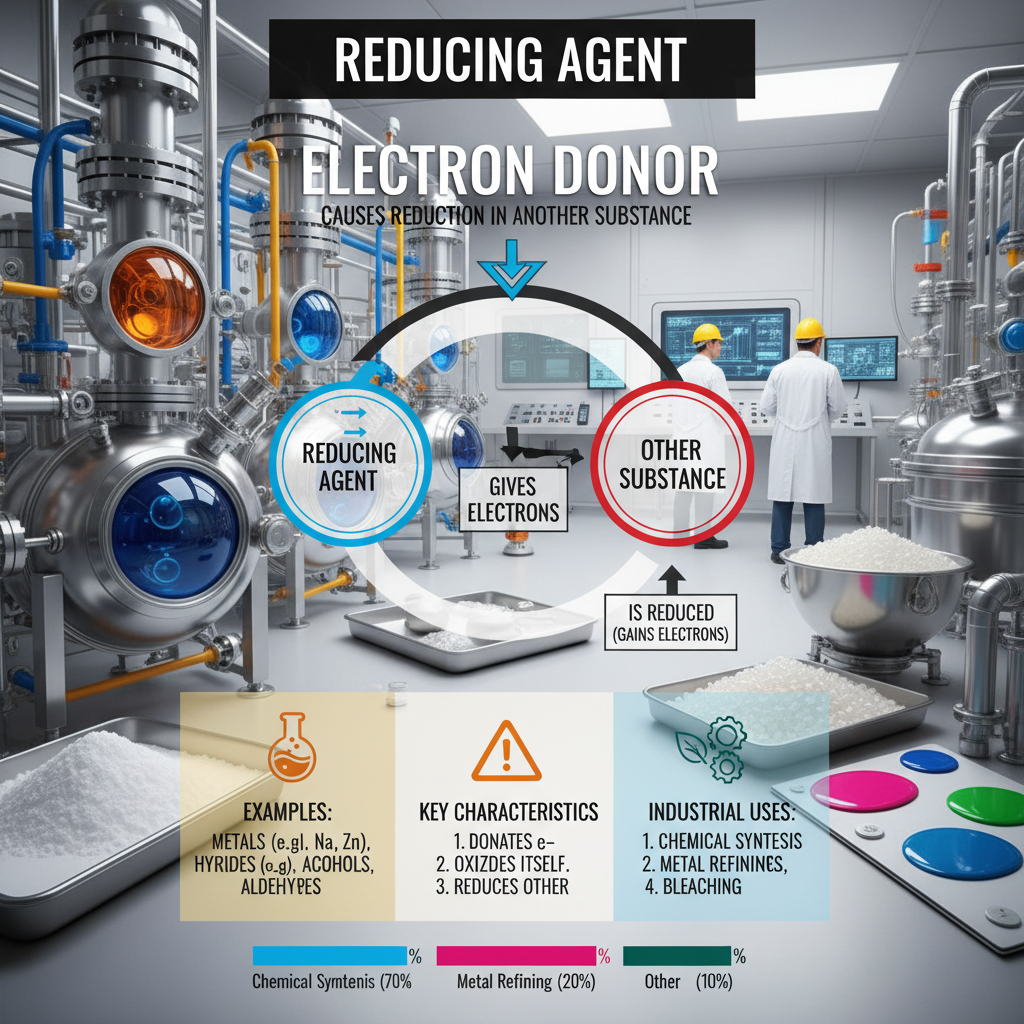
To accurately define reducing agent chemistry, one must look at the process of electron transfer. In the textile industry, a reducing agent is a substance that donates electrons to another molecule, effectively reducing the oxidation state of that molecule. This is most critical when dealing with sulfur dyes; the reducing agent converts the insoluble dye into a soluble "leuco" form, which can then migrate into the cotton or cellulose fiber.
The efficiency of this process depends heavily on the pH level and temperature of the dye bath. Sodium hydrosulfite, for example, is a common industry standard, but the precision of its application determines whether the fabric achieves a deep, uniform shade or suffers from spotting and uneven coloration. Understanding these molecular interactions is the first step toward achieving professional-grade textile finishing.
Industrial Significance and Global Standards
On a global scale, the demand for high-performance textiles has pushed the industry to refine how we define reducing agent protocols. According to industrial reports and ISO quality standards, the consistency of dyeing is directly linked to the purity and concentration of the reducing agents used. In hubs like Southeast Asia and India, where textile production is concentrated, the shift toward standardized chemical dosages has led to a significant decrease in fabric rejection rates.
One of the primary challenges addressed by modern reducing agents is the issue of "color bleeding" and instability. By employing highly stable reducing agents, manufacturers can ensure that the dye is locked into the fiber more securely. This not only improves the aesthetic quality of the garment but also extends its lifecycle, contributing to a more sustainable fashion ecosystem by reducing premature waste.
Furthermore, the integration of these chemicals into automated dosing systems has revolutionized the workshop floor. Digital transformation allows for real-time monitoring of the reduction potential in the dye bath, ensuring that the define reducing agent parameters are maintained within a narrow margin of error, which is essential for high-end luxury textile production.
Core Components for Maximum Efficacy
When we analyze the core components required to define reducing agent performance, durability of the chemical reaction is paramount. The agent must maintain its reducing power throughout the entire immersion cycle to prevent the dye from precipitating prematurely on the fabric surface, which would cause unfixable streaks.
Scalability is another critical factor; a reducing agent that works in a lab beaker must perform identically in a 10,000-liter industrial vat. This requires a high degree of solubility and compatibility with other auxiliaries, such as wetting agents and sequestering agents, to ensure that no antagonistic chemical reactions occur during the dyeing process.
Finally, cost-efficiency and safety cannot be overlooked. The modern industry seeks agents that provide maximum reduction potential with minimum dosage. This not only lowers the cost per kilogram of fabric but also reduces the chemical load in the effluent treatment plant (ETP), making the entire operation more eco-friendly and compliant with local environmental laws.
Practical Applications in Textile Dyeing
In real-world applications, the way we define reducing agent usage varies significantly between denim washing and traditional fabric dyeing. In denim production, reducing agents are often used in the "bleaching" or "stripping" phase to remove excess indigo from the seams and folds, creating that sought-after vintage look while maintaining the integrity of the cotton yarn.
Beyond denim, these agents are indispensable in the production of high-visibility safety wear and military uniforms, where deep blacks and navy blues are required. In these remote industrial zones, the use of stabilized reducing agents ensures that color depth is achieved even in varying water quality conditions, providing reliability across different geographic manufacturing sites.
Performance Analysis of Various Reducing Agent Types
Long-Term Value and Economic Benefits
The long-term value of utilizing high-grade reducing agents extends beyond the immediate color result. From a logical perspective, the reduction in "re-dyeing" costs is the most tangible benefit. When a factory can define reducing agent concentrations accurately, the first-time-right (FTR) percentage increases, dramatically cutting down on labor costs and energy consumption per batch.
From an emotional and brand-equity angle, the use of stabilized and safe reducing agents builds trust with international buyers. Brands today demand transparency in the supply chain; knowing that a manufacturer uses non-toxic, high-efficiency agents ensures the safety of the end consumer and the dignity of the workers handling the chemicals, fostering a relationship of innovation and mutual trust.
Future Trends in Green Reduction Technology
The future of how we define reducing agent technology is leaning heavily toward "Green Chemistry." We are seeing a transition from harsh inorganic salts to bio-based reducing agents derived from agricultural waste. These innovations aim to provide the same reduction potential as traditional hydrosulfites but with a fraction of the environmental footprint, making them biodegradable and easier to treat in wastewater.
Digital transformation is also playing a role through the advent of "Smart Dyeing" systems. These systems use AI to calculate the exact amount of reducing agent needed based on the fabric weight and dye concentration in real-time. This eliminates human error and ensures that not a single gram of chemical is wasted, aligning industrial production with global sustainability goals.
Moreover, the move toward closed-loop water systems means that reducing agents must be redesigned to be compatible with recycled water. Future agents will likely be designed to remain active even in the presence of residual salts from previous dyeing cycles, allowing factories to operate with zero liquid discharge (ZLD) without sacrificing quality.
Overcoming Common Challenges in Application
Despite the benefits, many factories struggle with the rapid decomposition of reducing agents, especially in hot and humid climates. When the agent decomposes before it can act on the dye, the result is patchy coloration. The expert solution to this is the use of "stabilized" formulations or the implementation of controlled-temperature storage facilities to maintain chemical potency.
Another common limitation is the high alkalinity often required for the reduction process, which can damage delicate fibers like silk or fine wool. To overcome this, the industry is developing "mild" reducing agents that function at a near-neutral pH. This innovation allows for the application of sulfur dyes to a wider range of fabric blends without compromising the tensile strength of the yarn.
Lastly, the management of sulfur-containing effluents remains a hurdle. By integrating advanced oxidation processes (AOPs) in the wastewater stage, factories can effectively neutralize the remnants of the reducing agents. This holistic approach—combining efficient application with advanced treatment—is the only way to maintain a competitive and compliant textile business.
Comparative Analysis of Reducing Agent Performance Across Fabric Types
| Agent Category | Fiber Compatibility | Reduction Speed | Eco-Impact Score (1-10) |
|---|---|---|---|
| Standard Hydrosulfite | Cotton / Linen | Very Fast | 4 |
| Stabilized Dithionite | Cotton Blends | Fast | 6 |
| Bio-Based Reducer | All Cellulosics | Moderate | 9 |
| Mild pH Agent | Silk / Wool / Cotton | Slow | 7 |
| Industrial Concentrate | Heavy Canvas | Very Fast | 5 |
| Enzymatic Reducer | Premium Cotton | Moderate | 10 |
FAQS
Defining the concentration requires calculating the total liquor ratio and the weight of the fabric. Generally, for sulfur dyes, a ratio of 2-5% based on the weight of the fabric is used, but this must be adjusted based on the dye's specific reduction potential and the water's hardness. We recommend a titration test to ensure the active content of the agent is sufficient for the specific shade required.
Rapid addition can lead to localized high concentrations, causing "spotting" or uneven dye penetration. The best practice is to dissolve the agent in a small amount of warm water first or use an automated dosing system that ensures a gradual, homogenous distribution across the dye bath to maintain consistency.
While sodium hydrosulfite is faster and more powerful, organic reducing agents are becoming highly competitive. They may require slightly longer immersion times or higher temperatures, but they offer superior fabric safety and a much lower environmental impact, making them the preferred choice for "Eco-Label" certified garments.
Reducing agents are highly sensitive to moisture and heat. They should be stored in airtight, moisture-proof containers in a cool, dry, and well-ventilated area. Avoid contact with any oxidizing agents or acids, as these can trigger premature decomposition and render the chemical ineffective for the dyeing process.
Yes, in principle, as both require a reduction to become soluble. However, the required pH levels and concentrations differ. Vat dyes typically require a more alkaline environment (using caustic soda) alongside the reducing agent, whereas sulfur dyes can often be processed at a slightly lower pH. Always check the technical data sheet for each dye type.
Excess reducing agents can increase the Chemical Oxygen Demand (COD) and Sulfide levels in the effluent. To manage this, we recommend a post-dyeing oxidation step (using hydrogen peroxide or air) to neutralize the remaining reducing power before the water enters the biological treatment stage of the ETP.
Conclusion
To effectively define reducing agent chemistry is to unlock the full potential of textile coloration. From the molecular electron transfer that makes dyes soluble to the industrial-scale application in denim and garment dyeing, these agents are the invisible force behind the vibrancy and durability of our clothes. By balancing reduction power with stability and environmental safety, manufacturers can achieve a superior product that meets both commercial and ecological standards.
Looking ahead, the integration of green chemistry and AI-driven dosing will redefine the industry, moving us toward a future of zero-waste textile production. For manufacturers seeking to optimize their processes, investing in high-purity, stabilized reducing agents is no longer an option but a necessity for survival in a competitive global market. We invite you to explore our full range of textile auxiliaries to elevate your production quality. Visit our website: www.dyeingchem.com
Related Posts
-

Exploring the Comprehensive Reducing Agent List for Industrial and Environmental Applications
-

Understanding the Crucial Role of List of Reducing Agents in Organic Chemistry
-
Exploring the Crucial Role of Oxidizing and Reducing Agents in Organic Chemistry
-

Understanding The Significance Of Both Oxidising And Reducing Agent In Modern Applications

William Lee
If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.




