Industrial Application of strong reducing agents organic chemistry
Table of Contents
In the complex landscape of chemical synthesis, the use of strong reducing agents organic chemistry plays a pivotal role in transforming functional groups and facilitating the creation of intricate molecular architectures. These powerful reagents are essential for converting carbonyls to alcohols, nitriles to amines, and esters to primary alcohols, providing the necessary thermodynamic drive to overcome significant energy barriers in organic reactions.
Globally, the demand for precision in chemical manufacturing—particularly in the production of pharmaceuticals, high-performance polymers, and textile auxiliaries—has elevated the importance of understanding these reagents. By mastering the selectivity and potency of strong reducing agents, chemists can optimize yield and reduce waste, aligning industrial output with the stringent requirements of modern quality standards and environmental regulations.
Understanding the nuances of strong reducing agents organic chemistry allows manufacturers to move beyond generic processes toward tailored chemical solutions. Whether it is in the development of specialized dyes or the synthesis of complex organic intermediates, the ability to control the reduction process is a cornerstone of innovation in the specialty chemicals sector, ensuring both product efficacy and process safety.

Fundamental Principles of Strong Reducing Agents

At its core, the mechanism of strong reducing agents organic chemistry involves the transfer of electrons or hydride ions to an electron-deficient center, typically a carbon atom in a carbonyl or nitrile group. Reagents such as Lithium Aluminum Hydride (LiAlH4) represent the pinnacle of this power, capable of reducing almost any carbonyl-containing compound to its corresponding alcohol or amine with high efficiency.
The effectiveness of these agents is determined by the nucleophilicity of the hydride source and the stability of the leaving group. In an industrial setting, choosing the right agent means balancing the need for total conversion with the necessity of avoiding over-reduction or unwanted side reactions, ensuring that the final chemical auxiliary maintains its intended functional properties.
Industrial Relevance and Global Market Impact
The global application of strong reducing agents organic chemistry is deeply intertwined with the growth of the specialty chemicals market. According to industry trends aligned with ISO standards for chemical safety and efficiency, the ability to synthesize high-purity organic intermediates is a primary driver of competitive advantage in the textile and leather auxiliary sectors.
One of the most significant challenges addressed by these reagents is the scalability of complex syntheses. In regions like Southeast Asia and China, where textile printing and dyeing industries are concentrated, the shift toward high-performance sulfur dyes and enzyme-based auxiliaries requires precise reduction steps to ensure color fastness and fabric integrity.
Furthermore, the integration of these agents into automated production lines has reduced human error and increased safety. By employing controlled-addition systems for highly reactive reducing agents, manufacturers can achieve consistent batch quality, reducing the environmental footprint associated with failed synthesis runs and hazardous waste.
Comparative Analysis of Reduction Potentials
When analyzing the spectrum of strong reducing agents organic chemistry, the primary differentiator is the reduction potential. While sodium borohydride is suitable for aldehydes and ketones, more aggressive tasks require the potency of metal hydrides or catalytic hydrogenation.
The strategic selection of strong reducing agents organic chemistry depends on the substrate's steric hindrance and the presence of other sensitive functional groups. For instance, the use of Diisobutylaluminum hydride (DIBAL-H) allows for the selective reduction of esters to aldehydes, a critical step in creating fragrance precursors for daily chemical auxiliaries.
Ultimately, the thermodynamic drive provided by these agents ensures that reactions proceed to completion even under mild conditions. This efficiency is paramount when dealing with heat-sensitive organic molecules, preventing the degradation of the product and ensuring a higher purity profile for final industrial use.
Performance Metrics in Textile Auxiliary Synthesis
In the production of textile and printing auxiliaries, the performance of a reducing agent is measured by its conversion rate and its impact on the final product's stability. The goal is to achieve maximum reduction of the target group while maintaining the structural integrity of the larger organic chain.
By utilizing specific strong reducing agents organic chemistry, manufacturers can fine-tune the hydrophilicity or lipophilicity of an auxiliary, which directly affects how the agent interacts with the fabric fiber during the dyeing process.
Efficiency Comparison of Reducing Agent Methods
Practical Application in Dyeing and Finishing
Within the realm of denim washing and sulfur dyeing, the application of strong reducing agents is essential for the "reduction clearing" process. This step removes unfixed dyes from the fabric surface, ensuring that the final garment has a clean, professional appearance and meets strict rub-fastness standards.
Beyond clearing, these agents are used in the synthesis of post-treatment auxiliaries that soften fabrics or provide water-repellent properties. By strategically reducing organic precursors, chemists can create molecules that bond more effectively to cellulose or protein fibers, enhancing the long-term durability of the textile finish.
Sustainability and Green Chemistry Integration
The evolution of strong reducing agents organic chemistry is now heavily focused on sustainability. Traditional reagents often produce significant salt waste or require hazardous solvents; however, the industry is moving toward catalytic systems that use hydrogen gas, which produces water as the only byproduct.
Green chemistry initiatives are also exploring the use of bio-based reducing agents and enzyme-catalyzed reductions. These innovations not only reduce the toxicity of the manufacturing process but also lower the energy requirements, as biological catalysts often operate at room temperature and atmospheric pressure.
For manufacturers of paper-making and oilfield auxiliaries, adopting these sustainable practices is no longer optional. Compliance with global environmental policies, such as REACH, necessitates a shift toward reagents that offer high selectivity with minimal ecological impact, ensuring the longevity of the industrial ecosystem.
Strategic Implementation Challenges and Solutions
One of the primary challenges in utilizing strong reducing agents is their extreme reactivity, which can lead to safety hazards if not managed correctly. Pyrophoric materials like LiAlH4 require inert atmospheres and specialized handling equipment to prevent spontaneous combustion upon contact with air or moisture.
To overcome these risks, many plants are implementing continuous flow chemistry. By processing small amounts of reagents in a controlled stream rather than in large batches, the potential impact of an accidental release is minimized, and heat dissipation is greatly improved, leading to safer and more consistent reactions.
Another hurdle is the cost of high-potency reagents. To address this, the industry is focusing on "reagent recycling" and the use of co-catalysts that allow for lower loading of the primary reducing agent. This strategic approach balances the need for chemical power with the economic necessity of cost-efficiency.
Comparative Analysis of Industrial Reducing Agents
| Reagent Type | Reduction Power | Safety Risk | Industrial Application |
|---|---|---|---|
| LiAlH4 | Extreme (10/10) | High (Pyrophoric) | Fine Chemical Synthesis |
| NaBH4 | Moderate (6/10) | Low (Stable) | General Dye Auxiliaries |
| DIBAL-H | High (9/10) | Medium (Reactive) | Selective Ester Reduction |
| H2/Pd Catalyst | High (8/10) | Medium (Pressure) | Mass Production/Green Chem |
| Zn/HCl | Low (4/10) | Low (Acidic) | Simple Organic Reductions |
| Sodium Dithionite | Moderate (5/10) | Low (Stable) | Sulfur Dye Reduction |
FAQS
LiAlH4 is a much stronger reducing agent capable of reducing esters, carboxylic acids, and amides, whereas NaBH4 is milder and typically only reduces aldehydes and ketones. In industrial textile auxiliary production, NaBH4 is preferred for safety and selectivity, while LiAlH4 is reserved for specialized high-potency syntheses.
Sulfur dyes are insoluble in water; strong reducing agents like sodium dithionite are used to reduce the dye to a soluble "leuco" form. This allows the dye to penetrate the textile fibers before it is re-oxidized to its original insoluble state, locking the color into the fabric.
Yes, catalytic hydrogenation using hydrogen gas and a metal catalyst (like Palladium or Nickel) is the primary eco-friendly alternative. It minimizes waste and eliminates the need for stoichiometric amounts of hazardous metal salts, aligning with modern green chemistry standards.
Handling requires an inert atmosphere (Nitrogen or Argon) to prevent reaction with oxygen and moisture. Personnel must use specialized PPE, including fire-resistant clothing and face shields, and work within blast-proof fume hoods to manage potential exothermic reactions.
A higher reduction potential generally ensures a faster and more complete conversion of the starting material. However, if the potential is too high, it can lead to over-reduction or the degradation of other functional groups, which actually lowers the yield of the desired target molecule.
Most strong reducing agents, like LiAlH4, react violently with water and must be used in anhydrous organic solvents. However, specific agents like sodium borohydride can be used in aqueous or alcoholic solutions, making them more suitable for certain daily chemical auxiliary applications.
Conclusion
The application of strong reducing agents organic chemistry is fundamental to the advancement of the specialty chemicals industry. From the precision required in textile auxiliary synthesis to the rigorous demands of sulfur dye processing, these reagents provide the chemical "muscle" needed to transform raw materials into high-value products. By balancing potency with selectivity and safety, manufacturers can ensure superior product quality and operational efficiency.
Looking forward, the integration of green chemistry and continuous flow technology will redefine how these powerful agents are utilized. The transition toward catalytic and bio-based reduction methods will not only reduce the environmental burden but also unlock new possibilities for molecular design. We encourage industry professionals to stay abreast of these innovations to maintain a competitive edge in the global market. Visit our website: www.dyeingchem.com
Related Posts
-
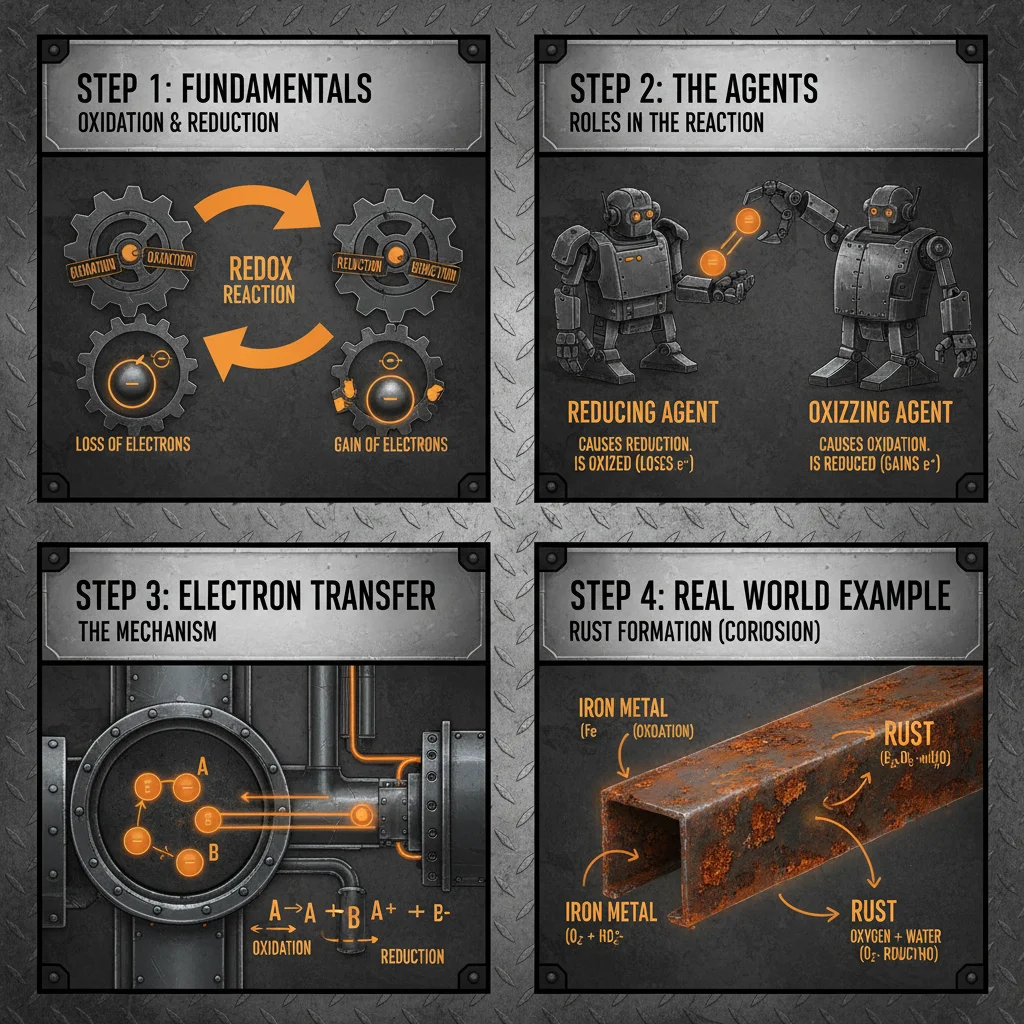
Understanding Reduction Agent and Oxidation Agent Processes and Their WideRanging Applications
-
Understanding Textile Auxiliaries Meaning for Optimized Textile Production
-

Leading Chemical Solutions for Advanced Industries Beyond
-
DTT Reducing Agent Applications in Biochemical Research and Pharmaceutical Industries

David Miller
If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.




