Understanding Reduction Agent and Oxidation Agent Processes and Their WideRanging Applications
Table of Contents
Reduction agent and oxidation agent are fundamental concepts in chemistry, with profound implications across numerous industries and scientific disciplines. These processes, at their core, involve the transfer of electrons between chemical species, driving reactions that power everything from energy production to the synthesis of vital materials. Understanding the principles behind reduction agent and oxidation agent is critical for innovation in areas like materials science, environmental remediation, and pharmaceutical development.
The global relevance of these concepts extends far beyond the laboratory. Industrial processes, such as metal refining, fertilizer production, and the creation of polymers, heavily rely on carefully controlled reduction agent and oxidation agent reactions. Furthermore, environmental concerns like water purification and pollution control demand a deep understanding of how these processes impact our planet. The demand for sustainable and efficient chemical processes continually drives research and development in this field.
Ultimately, grasping the nuances of reduction agent and oxidation agent empowers us to develop more effective technologies, address pressing environmental challenges, and improve the quality of life globally. The ability to manipulate these reactions allows for advancements in energy storage, medical treatments, and countless other applications, making it a cornerstone of modern scientific progress.
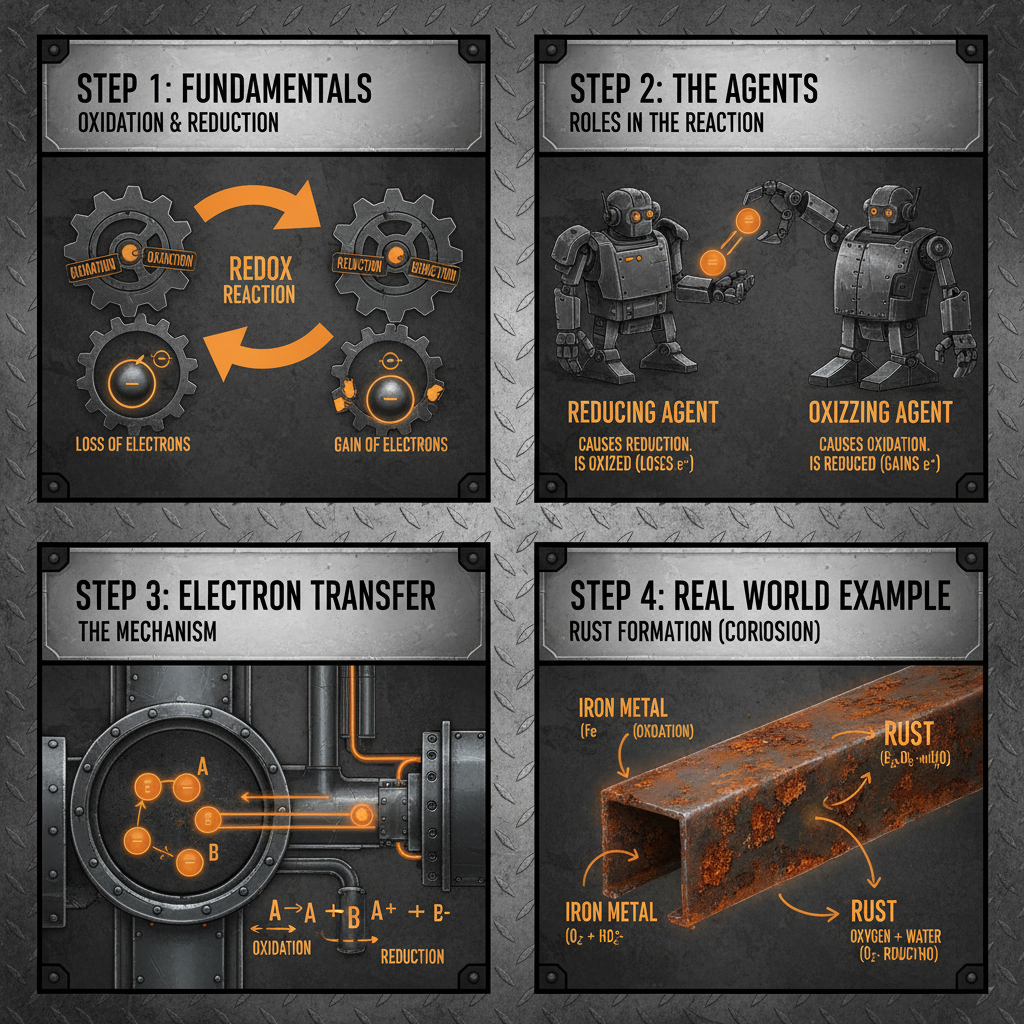
The Core Principles of reduction agent and oxidation agent

At its heart, a reduction agent and oxidation agent reaction involves the transfer of electrons. Reduction is the gain of electrons, while oxidation is the loss of electrons. These processes always occur simultaneously – one substance can't be reduced without another being oxidized. The substance that causes another to be oxidized is the reducing agent, and conversely, the substance that causes another to be reduced is the oxidizing agent.
These reactions are fundamental to countless processes, from the rusting of iron (oxidation) to the creation of energy in living cells (reduction-oxidation, or redox, reactions). The ability to control these electron transfers is key to many technological applications. The energy released or absorbed during reduction agent and oxidation agent reactions can be harnessed for power, or used to drive the synthesis of new compounds.
Global Industrial Significance of reduction agent and oxidation agent
The industrial relevance of reduction agent and oxidation agent is immense. The steel industry relies heavily on reduction agent and oxidation agent reactions to remove oxygen from iron ore, producing the steel essential for construction, manufacturing, and infrastructure. Similarly, the production of aluminum involves the reduction of aluminum oxide, a critical process for the aerospace, automotive, and packaging industries. These large-scale applications contribute significantly to the global economy.
Beyond metals, reduction agent and oxidation agent reactions are central to the production of countless chemicals, including fertilizers, plastics, and pharmaceuticals. The Haber-Bosch process, a crucial reduction agent and oxidation agent reaction, allows for the synthesis of ammonia, a key component of fertilizers, supporting global food production. This underscores the importance of these reactions in sustaining a growing global population.
Furthermore, the burgeoning field of battery technology is entirely dependent on reduction agent and oxidation agent reactions to store and release energy. The development of more efficient and sustainable batteries relies on innovative approaches to controlling these reactions, with far-reaching implications for electric vehicles and renewable energy storage.
Key Determinants of Efficient reduction agent and oxidation agent Reactions
Several factors govern the efficiency and selectivity of reduction agent and oxidation agent reactions. The choice of catalyst is paramount; catalysts lower the activation energy required for the reaction to proceed, accelerating the rate and improving yield. Different catalysts are suited for different reactions, and research into novel catalysts is an ongoing endeavor. The strength of the reducing or oxidizing agent also plays a critical role – a stronger agent will generally lead to a faster reaction.
Temperature and pressure are also significant determinants. Increasing the temperature generally increases the reaction rate, but can also lead to unwanted side reactions. Pressure can affect the equilibrium of gaseous reactions. Solvent choice can influence reaction rates and selectivity, often by stabilizing intermediate species or altering the solubility of reactants. Optimizing these parameters requires a thorough understanding of the specific reduction agent and oxidation agent reaction being studied.
Finally, the surface area of reactants in heterogeneous reactions (reactions involving different phases) is crucial. A larger surface area allows for more contact between reactants and catalyst, increasing the reaction rate. Nanomaterials, with their exceptionally high surface area to volume ratio, are increasingly being utilized to enhance reduction agent and oxidation agent processes.
Practical Applications and Industry Integration of reduction agent and oxidation agent
reduction agent and oxidation agent processes are woven into the fabric of modern life. In the food industry, antioxidants (reducing agents) are added to prevent spoilage and preserve freshness. Bleaching agents (oxidizing agents) are used in textile manufacturing and paper production to whiten materials. Water treatment plants employ reduction agent and oxidation agent reactions to disinfect water and remove pollutants.
The pharmaceutical industry relies heavily on reduction agent and oxidation agent chemistry for the synthesis of complex drug molecules. Many drugs undergo multiple reduction agent and oxidation agent steps during their production. Moreover, the development of new drugs often involves understanding how the drug interacts with biological systems through reduction agent and oxidation agent mechanisms.
Efficiency Comparison of reduction agent and oxidation agent Methods
reduction agent and oxidation agent in Environmental Sustainability
reduction agent and oxidation agent processes play a vital role in addressing environmental challenges. Advanced oxidation processes (AOPs) utilize powerful oxidizing agents to break down persistent organic pollutants in water and wastewater, offering a sustainable solution for water purification. Bioremediation techniques leverage the natural reduction agent and oxidation agent capabilities of microorganisms to clean up contaminated soil and water.
Furthermore, the development of green chemistry principles emphasizes the use of environmentally friendly reduction agent and oxidation agent reactions that minimize waste and reduce the use of hazardous chemicals. Electrocatalytic reduction agent and oxidation agent reactions, powered by renewable energy sources, offer a promising avenue for sustainable chemical synthesis and pollution control.
Advantages and Future Projections of reduction agent and oxidation agent
The advantages of mastering reduction agent and oxidation agent reactions are multifaceted: increased efficiency in industrial processes, the development of innovative materials, and solutions to critical environmental problems. The ability to control these reactions enables the creation of products with enhanced properties and reduced environmental impact.
Future projections point toward a greater emphasis on sustainable and efficient reduction agent and oxidation agent processes. The development of new catalysts, powered by artificial intelligence and machine learning, will accelerate the discovery of more selective and active catalysts. Integration with renewable energy sources will further reduce the carbon footprint of these processes, promoting a circular economy.
The exploration of novel redox materials, such as metal-organic frameworks (MOFs) and carbon nanomaterials, promises to unlock new possibilities in energy storage, catalysis, and sensing. These advancements will contribute to a more sustainable and technologically advanced future.
Challenges and Innovative Solutions in reduction agent and oxidation agent Application
Despite significant advancements, challenges remain in the application of reduction agent and oxidation agent reactions. Controlling selectivity and minimizing unwanted side reactions can be difficult, requiring precise optimization of reaction conditions. The use of hazardous chemicals in some processes poses environmental and safety concerns. The cost of certain catalysts and reagents can also be prohibitive.
Innovative solutions are being developed to address these challenges. Flow chemistry, which involves carrying out reactions in continuous flow reactors, offers improved control over reaction parameters and reduced waste generation. The development of earth-abundant and non-toxic catalysts is gaining momentum. Electrochemistry offers a greener alternative to traditional chemical reducing and oxidizing agents.
The use of computational modeling and machine learning is accelerating the discovery of new catalysts and reaction pathways, enabling the design of more efficient and sustainable reduction agent and oxidation agent processes. These advancements are paving the way for a more responsible and environmentally conscious chemical industry.
Summary of Current Challenges and Corresponding Solutions in reduction agent and oxidation agent Application
| Challenge | Impact on Efficiency | Proposed Solution | Expected Improvement |
|---|---|---|---|
| Low Selectivity | Increased By-products | Development of Tailored Catalysts | 80-90% Selectivity |
| Hazardous Reagents | Environmental Pollution | Electrochemical Reduction/Oxidation | Reduced Waste |
| High Catalyst Cost | Reduced Economic Viability | Earth-Abundant Metal Catalysts | Cost Reduction by 50% |
| Poor Mass Transfer | Slow Reaction Rates | Microreactors/Flow Chemistry | Enhanced Reaction Rate |
| Difficult Reaction Control | Inconsistent Product Quality | Automated Process Control | Improved Product Consistency |
| Energy Intensive Processes | High Carbon Footprint | Photocatalysis | Reduced Energy Consumption |
FAQS
reduction agent and oxidation agent reactions are fundamental to numerous industrial processes, including metal refining, fertilizer production, polymer synthesis, and the manufacturing of pharmaceuticals. They are utilized for everything from extracting metals from ores to creating complex organic molecules. Understanding and controlling these reactions is essential for optimizing efficiency and producing high-quality products. The field is continually evolving to prioritize sustainability and minimize environmental impact.
Identifying reduction agent and oxidation agent reactions involves looking for changes in oxidation states of the involved elements. If an element's oxidation state decreases, it's being reduced (gain of electrons). If it increases, it's being oxidized (loss of electrons). Indicators include the transfer of oxygen, hydrogen, or electrons, and changes in color or the release of energy (often as heat or light).
Traditional reduction agent and oxidation agent processes can generate significant waste products, including hazardous chemicals and greenhouse gases. The use of toxic oxidizing and reducing agents poses a risk to environmental and human health. Energy-intensive processes contribute to carbon emissions. These concerns are driving the development of greener alternatives, such as electrochemistry and photocatalysis.
Catalysts accelerate reduction agent and oxidation agent reactions without being consumed in the process. They lower the activation energy required for the reaction to occur, increasing the reaction rate and improving selectivity. Different catalysts are suited for different reactions, and the choice of catalyst is crucial for optimizing process efficiency. Research into novel catalysts is a major area of focus in chemical research.
Future trends include the development of sustainable and efficient catalysts, integration with renewable energy sources, and the use of artificial intelligence and machine learning to accelerate the discovery of new reaction pathways. Electrocatalysis, photocatalysis, and flow chemistry are expected to play increasingly important roles in greener and more sustainable chemical processes.
reduction agent and oxidation agent reactions are fundamental to battery technology, enabling the storage and release of electrical energy. The efficiency and capacity of batteries are directly related to the kinetics and thermodynamics of the redox reactions occurring within them. Ongoing research focuses on developing new electrode materials and electrolytes to enhance battery performance and create more sustainable energy storage solutions.
Conclusion
In summary, understanding reduction agent and oxidation agent is paramount to grasping a wide range of scientific and industrial processes. From fundamental chemical principles to global industrial applications and environmental sustainability, these reactions shape our world. The ability to control these processes allows for advancements in energy production, material science, and environmental remediation.
Looking ahead, continued research and innovation in reduction agent and oxidation agent chemistry are crucial for addressing pressing global challenges. The development of sustainable catalysts, integration with renewable energy sources, and the application of advanced computational techniques will pave the way for a more efficient, environmentally responsible, and technologically advanced future. Visit our website at www.dyeingchem.com to learn more about reduction agent and oxidation agent and their applications.
Related Posts
-

Exploring the Versatile Applications of Inorganic Reducing Agents in Industry and Beyond
-

The Vital Role of Good Reducing Agents in Modern Industries
-
Advanced Polymer Auxiliaries Chemicals for Enhanced Industrial Performance
-
Navigating High Performance Concrete Admixtures and textile auxiliaries manufacturer Solutions

Daniel Garcia
If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.




